Phendimetrazine
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Bontril |
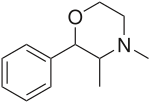
| Other names | Mephenmetrazine; (2S,3S)-3,4-Dimethyl-2-phenylmorpholine |
| AHFS/Drugs.com | Monograph |
| Routes of administration | By mouth |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | Peak plasma levels occur within 1 to 3 hours. Absorption is usually complete by 4 to 6 hours |
| Metabolism | Liver |
| Elimination half-life | 19-24 hours |
| Excretion | Urinary elimination |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.010.186 |
| Chemical and physical data | |
| Formula | C12H17NO |
| Molar mass | 191.274 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Phendimetrazine is a central nervous system (CNS) stimulant and anorectic drug of the morpholine chemical class, as well as a prodrug of phenmetrazine, approved for medical use as an anti-obesity drug and appetite suppressant, indicated as a second-line, short-term treatment of exogenous obesity,[2]
Originally marketed under the brand name Bontril, later amended to Bontril PDM to distinguish it from the time-released combination drug product line, Bontril Timed, which was an already-established, and unrelated product formulation with different active ingredients. Upon the expiration of Bontril's drug patent, generic drug manufacturers and some small-scale laboratories (e.g. Tutag Pharmaceuticals of New Jersey; Smith, Kline & French; Roerig Laboratories; Irwin, Neisler & Co.) released generic products under trade names including Prelu-2 and Adipost, Appecon, Melfiat, Obezine Phendiet, Plegine, Statobex, and TrimTabs.
Pharmacology
[edit]Mechanism of Action
[edit]Phendimetrazine is a prodrug of phenmetrazine (at the time, a well-known diet pill branded Preludin, meaning the former substance converts or metabolizes into, and produces, the latter substance upon ingestion, metabolization process occurs. Phenmetrazine (Preludin) is a substituted amphetamine developed and derived from amphetamine in the early 1950s and introduced to the market by the late 1950s. It was frequently prescribed as a weight loss medication from shortly after its released, and was discontinued in 1982 amidst growing awareness of the drug's widespread misuse, abuse and diversion. As with any prodrug, the conversion process is bound by the timeframe of the process of drug metabolism, hence it is effectively an extended-release formulation of a relatively low dose of phenmetrazine; consequently having a lower abuse potential. Phendimetrazine is an anorectic drug which acts as a norepinephrine-dopamine releasing agent (NDRA).[3]
Chemical and Molecular Structure
[edit]As an amphetamine congener, its structure incorporates the backbone of methamphetamine, a potent central nervous system stimulant. While the addition of an N-methyl group to amphetamine significantly increases its potency and bioavailability, methylation of phenmetrazine renders the compound virtually inactive. However, phendimetrazine is a prodrug for phenmetrazine which acts as the active metabolite. Phendimetrazine possesses preferable pharmacokinetics over phenmetrazine as a therapeutic agent because its metabolization by demethylases produces a more steady and prolonged exposure of active drug within the body. This decreases abuse potential as the peak blood-concentration of active phenmetrazine that's produced from a single dose of phendimetrazine is lower than a single therapeutically equivalent dose of phenmetrazine.
Chemical Synthesis
[edit]The reaction between N-methylethanolamine and 2-bromopropiophenone gives compound (3), which is reductively cyclized using formic acid to synthesize phendimetrazine.[4][5]
Dosage Forms and Directions for Use
[edit]Phendimetrazine is an oral medicine produced in the dosage form of a tablet or capsule, with each "pill" containing 35mg phendimetrazine as an active ingredient, with directions for use to ingest one 35mg tablet 45-60 minutes before meals, but not exceeding three doses daily. The extended-release formulation Anorex-SR was dosed one time daily (upon waking or as early as necessary, containing an aggregate 105mg of phendimetrazine in a long-acting, sustained-release formulation, providing a steady, slow release of phendimetrazine and 30% active metabolite, phenmetrazine, through the day. Anorex-SR containing in aggregate 105mg of the substance in the dosage form of a for once daily dosing, typically consumed 30 to 60 minutes before a morning meal. Whereas the immediate-release formulation has a maximum daily dosage of 210mg (6 tablets), the extended-release capsules have a maximum daily dosage of 105mg (one capsule).
Legal Regulation, Designation as a Controlled Substance
[edit]According to the List of Psychotropic Substances under International Control published by the International Narcotics Control Board, phendimetrazine is a Schedule III controlled substance under the Convention on Psychotropic Substances.[6] At the national level, U.S. correlates to the convention's ruling. The Controlled Substances Act of 1970 classifies the substance as Schedule III, alongside many anabolic steroids.
References
[edit]- ^ Anvisa (31 March 2023). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 4 April 2023). Archived from the original on 3 August 2023. Retrieved 16 August 2023.
- ^ Landau D, Jackson J, Gonzalez G (2008). "A case of demand ischemia from phendimetrazine". Cases J. 1 (1): 105. doi:10.1186/1757-1626-1-105. PMC 2531092. PMID 18710555.
- ^ Rothman RB, Baumann MH (2006). "Therapeutic potential of monoamine transporter substrates". Current Topics in Medicinal Chemistry. 6 (17): 1845–59. doi:10.2174/156802606778249766. PMID 17017961. Retrieved 5 May 2020.
- ^ "Phendimetrazine". Thieme. Retrieved 30 June 2024.
- ^ Werner Heel and Karl Zeile, U.S. patent 2,997,469 (1961 to Ingelheim, Germany, assignors to C. H. Boehringer Sohn, Ingelheim, Germany, a partnership).
- ^ "List of psychotropic substances under international control" (PDF). Archived from the original (PDF) on 31 August 2012. Retrieved 15 June 2005.
Pharmacology, Chemistry, and Synthesis
[edit]Mechanism of Action
[edit]Phendimetrazine is a prodrug of phenmetrazine (at the time, a well-known diet pill branded Preludin, meaning the former substance converts or metabolizes into, and produces, the latter substance upon ingestion, metabolization process occurs. Phenmetrazine (Preludin) is a substituted amphetamine developed and derived from amphetamine in the early 1950s and introduced to the market by the late 1950s. It was frequently prescribed as a weight loss medication from shortly after its released, and was discontinued in 1982 amidst growing awareness of the drug's widespread misuse, abuse and diversion. As with any prodrug, the conversion process is bound by the timeframe of the process of drug metabolism, hence it is effectively an extended-release formulation of a relatively low dose of phenmetrazine; consequently having a lower abuse potential. Phendimetrazine is an anorectic drug which acts as a norepinephrine-dopamine releasing agent (NDRA).[1]
Molecular Structure
[edit]As an amphetamine congener, its structure incorporates the backbone of methamphetamine, a potent central nervous system stimulant. While the addition of an N-methyl group to amphetamine significantly increases its potency and bioavailability, methylation of phenmetrazine renders the compound virtually inactive. However, phendimetrazine is a prodrug for phenmetrazine which acts as the active metabolite. Phendimetrazine possesses preferable pharmacokinetics over phenmetrazine as a therapeutic agent because its metabolization by demethylases produces a more steady and prolonged exposure of active drug within the body. This decreases abuse potential as the peak blood-concentration of active phenmetrazine that's produced from a single dose of phendimetrazine is lower than a single therapeutically equivalent dose of phenmetrazine.
Chemistry and Synthesis
[edit]The reaction between N-methylethanolamine and 2-bromopropiophenone gives compound (3), which is reductively cyclized using formic acid to synthesize phendimetrazine.[2][3]
Dosage Forms and Directions for Use
[edit]Phendimetrazine is an oral medicine produced in the dosage form of a tablet or capsule (colloquially a "pill"), with each containing of 35mg phendimetrazine as the active ingredient, with directions for use to ingest one 35mg tablet 45 minutes to an hour before meals, up to, but not exceeding, three doses daily. The extended-release formulation Anorex-SR was taken one time daily, upon waking or as early as needed, and contained 105mg of the active ingredient in modified-release dosage form, providing controlled, sustained release of the drug throughout the day. Phendimetrazine was previously manufactured as Anorex-SR, containing in aggregate 105mg of the substance in the dosage form of a Sustained-Release capsule for once daily dosing, typically consumed 30 to 60 minutes before a morning meal. Whereas the immediate-release formulation has a maximum daily dosage of 210mg (6 tablets), the extended-release capsules have a maximum daily dosage of 105mg (one capsule).
Legality, Regulation, International Controlled Substance Designation
[edit]According to the List of Psychotropic Substances under International Control published by the International Narcotics Control Board, phendimetrazine is a Schedule III controlled substance under the Convention on Psychotropic Substances.[4] At the national level, U.S. correlates to the convention's ruling. The Controlled Substances Act of 1970 classifies the substance as Schedule III, alongside many anabolic steroids.
References
[edit]- ^ Rothman RB, Baumann MH (2006). "Therapeutic potential of monoamine transporter substrates". Current Topics in Medicinal Chemistry. 6 (17): 1845–59. doi:10.2174/156802606778249766. PMID 17017961. Retrieved 5 May 2020.
- ^ "Phendimetrazine". Thieme. Retrieved 30 June 2024.
- ^ Werner Heel and Karl Zeile, U.S. patent 2,997,469 (1961 to Ingelheim, Germany, assignors to C. H. Boehringer Sohn, Ingelheim, Germany, a partnership).
- ^ "List of psychotropic substances under international control" (PDF). Archived from the original (PDF) on 31 August 2012. Retrieved 15 June 2005.

